TIL therapy, tumor-infiltrating lymphocyte therapy, is a type of adoptive cell immunotherapy that uses T cells taken directly from a patient’s tumor, expands them outside the body and then reinfuses them to help attack cancer.
It is especially relevant in solid tumors because it uses lymphocytes that have already reached the tumor environment and may already recognize tumor-specific targets. That makes TIL therapy one of the most interesting cell-therapy approaches for cancers where conventional options may be limited.
TIL therapy uses the patient’s own tumor-reactive immune cells and turns them into a larger, more active therapeutic product through controlled expansion.
What is TIL therapy?
TIL therapy is an autologous cell therapy based on tumor-infiltrating lymphocytes, which are T cells already present inside a patient’s tumor tissue. These cells are collected from the tumor, selected and expanded in the laboratory, then returned to the patient as a therapeutic infusion.
The rationale is simple but powerful: if some T cells have already found the tumor, expanding those cells to very large numbers may strengthen the immune attack in a more direct way than relying only on the natural immune response.
TIL therapy does not start from generic immune cells. It starts from lymphocytes that were already inside the tumor.
How TIL therapy works
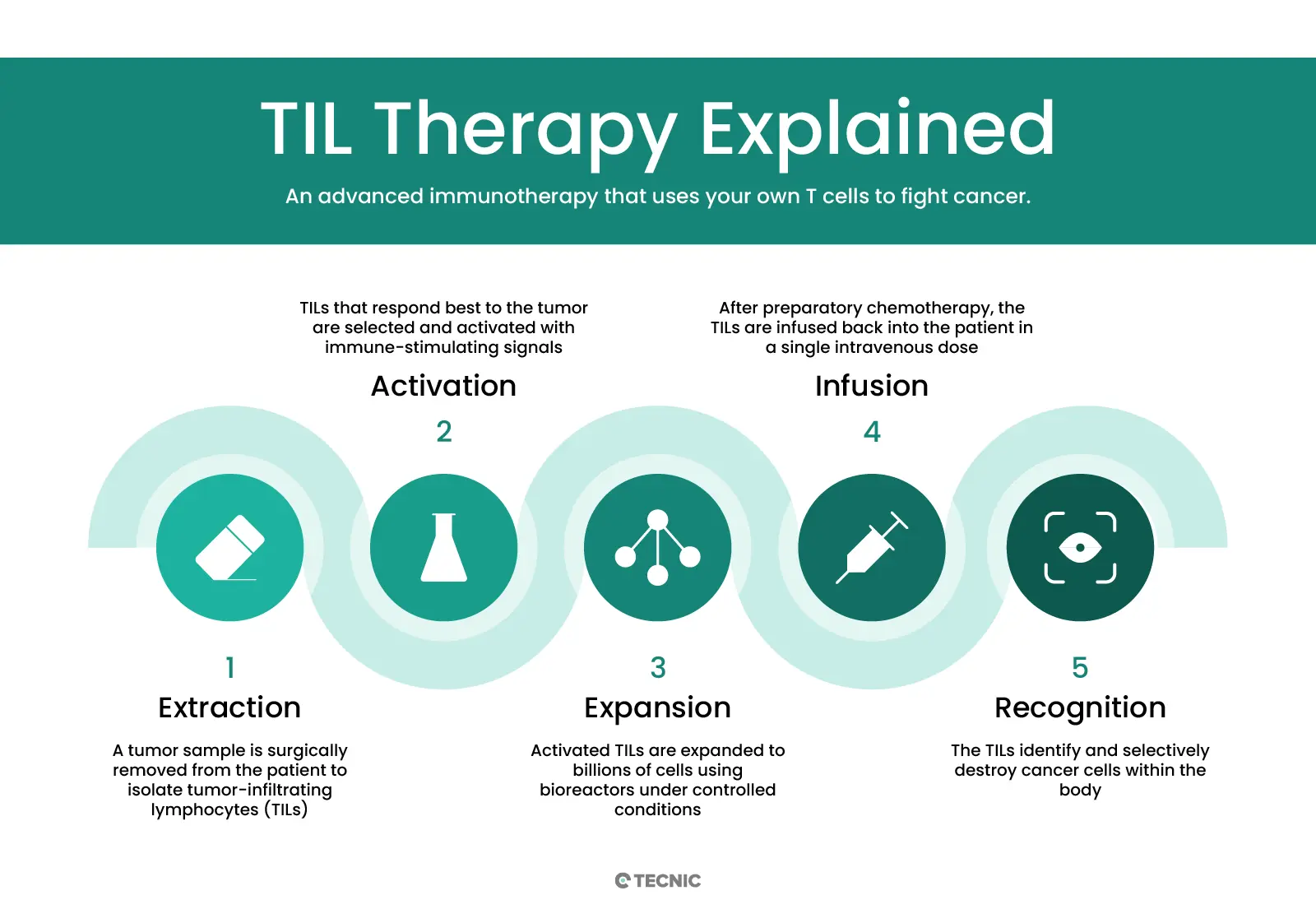
The treatment usually follows a defined sequence from tumor sampling to reinfusion. While protocols can vary, the logic is consistent across most TIL workflows.
A tumor sample is obtained surgically so the infiltrating lymphocytes can be isolated.
TIL populations are recovered from the tumor tissue and prepared for ex vivo culture.
The cells are expanded over several weeks, often with IL-2 support, until they reach therapeutic scale.
The expanded TIL product is infused back into the patient after a lymphodepleting preparation regimen.
TIL therapy is not only an immunology concept, it is also a demanding cell-manufacturing process.
Why TIL therapy matters for solid tumours
TIL therapy has attracted so much attention because solid tumors have often been harder to treat with cell therapy than hematological cancers. TILs offer a different route by starting with lymphocytes that have already infiltrated the tumor microenvironment.
Natural tumour recognition
TILs may already recognize multiple tumor-associated targets, which can help them address tumor heterogeneity better than a single-target approach.
Relevance in solid tumours
Because the cells come from the tumor site itself, they are especially relevant in solid-tumor immunotherapy strategies.

Main benefits and limitations of TIL therapy
TIL therapy offers clear advantages, but it also remains one of the most demanding autologous cell-therapy workflows in oncology.
TIL therapy is scientifically powerful, but its full impact depends on solving manufacturing, logistics and scale-up challenges.
Manufacturing and cell expansion in TIL therapy
One of the most important parts of TIL therapy is the expansion phase. The cells must grow from a small tumor-derived starting population into billions of cells while maintaining viability, functionality and clinical suitability.
That is why closed systems, reproducible culture conditions, monitoring and scalable bioreactor strategies are becoming increasingly important in TIL manufacturing. The therapy is personalized, but the process still needs industrial-level control.

How TECNIC fits this workflow
TECNIC fits this topic directly because TIL therapy depends on controlled cell expansion, robust monitoring and scalable bioprocess environments. As TIL workflows move from experimental programs toward broader clinical use, the quality of the expansion platform becomes increasingly important.
Bioreactors
Relevant when TIL and other advanced cell therapies need controlled expansion from early development to larger manufacturing workflows.
Cell and gene therapy context
TIL therapy fits naturally within the broader advanced-therapy field already reflected in TECNIC content.
Software and control
Cell therapy manufacturing benefits from stronger traceability, user control and repeatable process execution.
Contact TECNIC
When advanced cell therapy workflows need stronger expansion control and scale-up logic, direct technical discussion becomes more useful than theory alone.
This article works best when TIL therapy is framed as both a clinical immunotherapy and a high-control cell-manufacturing process.
Frequently asked questions
What is TIL therapy?
It is a form of adoptive cell therapy that uses tumor-infiltrating lymphocytes taken from the patient’s own tumor.
How does TIL therapy work?
T cells are isolated from the tumor, expanded in the lab and reinfused into the patient after a conditioning regimen.
Why is TIL therapy important for solid tumours?
Because it starts from immune cells that have already reached the tumor site and may already recognize tumor-specific targets.
Is TIL therapy the same as CAR-T therapy?
No. TIL therapy expands naturally tumor-infiltrating T cells, while CAR-T therapy genetically engineers blood-derived T cells to express a designed receptor.
What is the biggest challenge in TIL therapy?
One of the biggest challenges is manufacturing, especially scaling up personalized cell expansion while maintaining quality and consistency.
Exploring how TIL therapy connects with scalable cell expansion and process control?
Explore TECNIC’s bioprocess solutions or speak with our team to review the right setup for advanced cell-therapy manufacturing workflows.